Nitric Acid Assisted In Situ Generation of BrOH: A Selective Catalyst for Oxidation of Benzylic Alcohols
Venkatanarayana Pappula and Subbarayappa Adimurthy
DOI10.21767/2472-1123.100024
Venkatanarayana Pappula and Subbarayappa Adimurthy*
Academy of Scientific and Innovative Research, CSIR - Central Salt and Marine Chemicals Research Institute, GB Marg, Bhavnagar, Gujarat, India
- *Corresponding Author:
- Subbarayappa Adimurthy
Academy of Scientific and Innovative
Research, CSIR - Central Salt and Marine
Chemicals Research Institute, GB Marg
Bhavnagar-364 002, Gujarat, India
Tel: 2567760
E-mail: adimurthy@csmcri.org
Received date: August 07, 2017; Accepted date: September 05, 2017; Published date: September 12, 2017
Citation: Pappula V, Adimurthy S. Nitric Acid Assisted In Situ Generation of BrOH: A Selective Catalyst for Oxidation of Benzylic Alcohols. J Org Inorg Chem Vol. 3 No. 2:4. doi:10.21767/2472-1123.100024
Abstract
Nitric acid (15 mol%) assisted in-situ generated hypobromous acid catalyzed aerobic oxidation of benzyl alcohols to aldehydes under metal-free aqueous conditions is described. Nitric acid plays a dual role as “H+” donor as well as secondary oxidant to convert bromide to a reactive species (BrOH) in presence of molecular oxygen from air.
Keywords
Benzylic alcohols; Oxidation; Nitric acid; Bromide-bromate; Benzaldehydes
Introduction
Oxidation of alcohols to carbonyl compounds is one of the most important and fundamental functional group transformations in synthetic organic chemistry [1]. To date many excellent catalytic methods have been developed for oxidation of benzylic alcohols [2-5]. Transition metal or nitroxyl radical catalysed oxidation of alcohols to aldehydes or ketones has attracted due to the use of molecular oxygen or H2O2 as terminal oxidants [6-11]. Alternate to metal catalysts, halogen species (Cl, Br, and I) are frequently used for the oxidation of alcohols [12,13]. Numerous methods have been reported for the oxidation of alcohols with varying amount (2-40 mol%) of bromine or bromine based catalysts [14-22]. One of cost-effective oxidizing reagent is chlorine-gas/hypochlorite but its direct use in organic synthesis is limited due to the possibility of non-selective oxidations [23-26]. Additionally, there are useful halogen or halide derivatives such as oxy-halo compounds of bromine and iodine [27-33]. and N-halosuccinimides [34]. are reported for the oxidation of alcohols. Oxone has been attractive oxidant and offers several advantages such as non-toxic nature, controlled addition, stability, easy handling and ease of transport [35]. However, moleculer oxygen and aqueous H2O2 are greener and atom economical oxidant than oxone [36,37]. Most of the alcohol oxidation protocols were based on transition metal catalysts, these methods suffer from multiple limitations, such as requires specific conditions like inert reaction atmosphere, high temperature, toxic nature of catalyst, high price of precious metals, use of co-catalyst and reaction disposal has low environmental acceptance. On the other hand, the methods avoiding transition metal catalysts requires stoichiometric or more than stoichiometric amount of halogen sources along with other additives as well as co-catalysts [38-40].
Experimental Section
General
All commercially available chemicals and reagents were used without any further purification unless otherwise indicated. 1H and 13C NMR spectra were recorded at 200 and 125 MHz, respectively. The spectra were recorded in CDCl3 as solvent. Multiplicity was indicated as follows: s (singlet); d (doublet); t (triplet); m (multiplet); dd (doublet of doublets), sp (septet) etc. and coupling constants (J) were given in Hz. Chemical shifts are reported in ppm relative to TMS as an internal standard. The peaks around delta values of 1H NMR (7.2), and 13C NMR (77) are correspond to deuterated solvent CDCl3. Progress of the reactions was monitored by thin layer chromatography (TLC). All products were purified through column chromatography using silica gel 100-200 mesh size using hexane. All the compounds were known and spectra is matched with reported literature.
General experimental procedure for the oxidation of benly alcohol to benzaldehyde (2a)
In a 25 mL round bottomed flask, 2 mmol of benzyl alcohol (1a), 10 mol% of NaBr/NaBrO3 5:1 couple and 2 ml of dioxane/ water (10:1) were placed. To this reaction flask 15 mol% of HNO3 (27 mg) added. The reaction flask was placed in an oil bath at 80°C connected with condenser and continued stirring for 12 hours, after completion of the reaction mixture allowed to room temperature then extracted with 3 × 25 ml ethyl acetate and dried over anhydrous sodium sulphate. Removal of the solvent under reduced pressure, the residue left out was subjected to column chromatography on silica gel (200-400) and hexane/ethyl acetate as eluent resulted in 62% isolated yield of benzaldehyde.
Results and Discussion
In our previous communication [41,42], we disclosed an efficient and selective oxidation of benzylic/secondary alcohols to the corresponding aldehydes/ketones with a catalytic amount of bromide-bromate (10 mol %) couple as “Br” source, H2O2 as stoichiometric oxidant (1.2 eq.) and H+-acid as an activator (15 mol %) at room temperature. The bromide-bromate couple in presence of acid generate BrOH/Br2 in-situ which was also supported from UV-vis study [41,42]. However, there is strong need for more efficient, selective and sustainable methods that avoids the use of specific and harsh reaction conditions, stoichiometric reagents, additives, and heavy metals as catalysts for alcohol oxidations. In continuation of our interest to develop sustainable and economical processes [43-49]. We hypothesize to replace the stoichiometric use of H2O2 with catalytic oxidants that would be further easily reoxidized and recycled by utilising atmospheric oxygen as secondary stoichiometric oxidant without any metal catalyst. To our delight easily available nitric acid was found to be suitable for such oxidations. Our present investigation for the oxidation of alcohols to aldehydes is guided by the generation of reactive species depicted in the following chemical equations (eqns. 1-7).
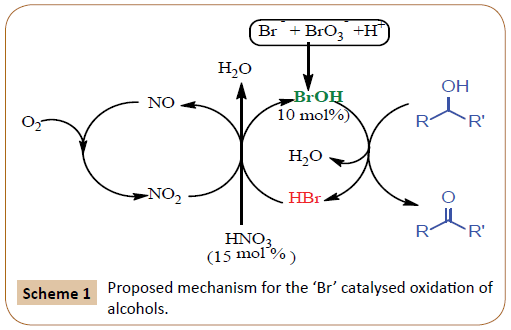
In present communication we realized that catalytic amount (15 mol %) of nitric acid plays dual role as a “H+” donor (eqn. 1 and 2) to activate the bromide-bromate couple as well as secondary oxidant to regenerate the active bromide species (eqs. 3 and 4) at mild temperature (80°C). Further, HBr is also oxidized by NO2 to generate BrOH/Br2 and NO (eqn. 5). In the next step, atmospheric oxygen oxidizes NO to NO2 species (eqn. 6) which upon water hydrolysis re-generate HNO3 (eqn. 7). In this way the BrOH/Br2 oxidants are effectively regenerated and reutilized for aerobic oxidation of alcohols.
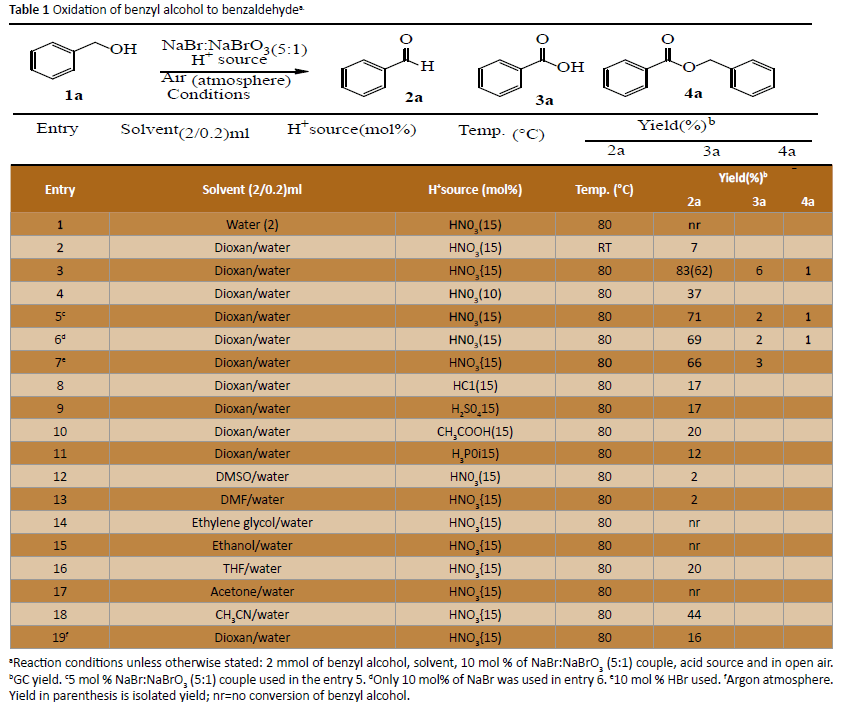
To optimise suitable reaction conditions, we performed the various experiments using benzyl alcohol, bromide-bromate couple and nitric acid as model reaction parameters under various reaction conditions (Table 1). Initially, the reaction was conducted in water as solvent at 80°C (entry 1), and only 7% of the yield was obtained in dioxane-water mixture at room temperature with 10 mol % bromide-bromate couple as source of Br2/BrOH, nitric acid (15 mol %) as a proton “H+” source (entry 2). It was observed that the reaction has considerable dependence on the solvent, temperature and amount of catalyst (Table 1, entry 3). When the concentration of nitric acid was decreased, the yield of 2a was dropped to 37% (Table 1, entry 4).
aReaction conditions unless otherwise stated: 2 mmol of benzyl alcohol, solvent, 10 mol % of NaBr:NaBrO3 (5:1) couple, acid source and in open air.bGC yield. c5 mol % NaBr:NaBrO3 (5:1) couple used in the entry 5. dOnly 10 mol% of NaBr was used in entry 6. e10 mol % HBr used. fArgon atmosphere. Yield in parenthesis is isolated yield; nr=no conversion of benzyl alcohol.
Figure 1:Oxidation of benzyl alcohol to benzaldehydea.
While decreasing the total bromide concentration to 5 mol% from 15 mol%, yield was also decreased (Table 1, entry 5). Performing the reaction either with NaBr or HBr alone (without bromate) the oxidation was not efficient (Table 1, entries 6 and 7). The reaction was not effective with other acids (HCl, H2SO4, CH3CO2H, H3PO4) and different solvents screened (Table 1, entries 8-18). Nitric acid has been largely studied as an oxidizing agent either in concentrated acid as solvent media [50-52]. Oxidation reaction under argon atmosphere was also not successful. Therefore, these parameters (Table 1, entry 2) were set as optimum for the further oxidation of benzylic alcohols to aldehydes (Table 2).
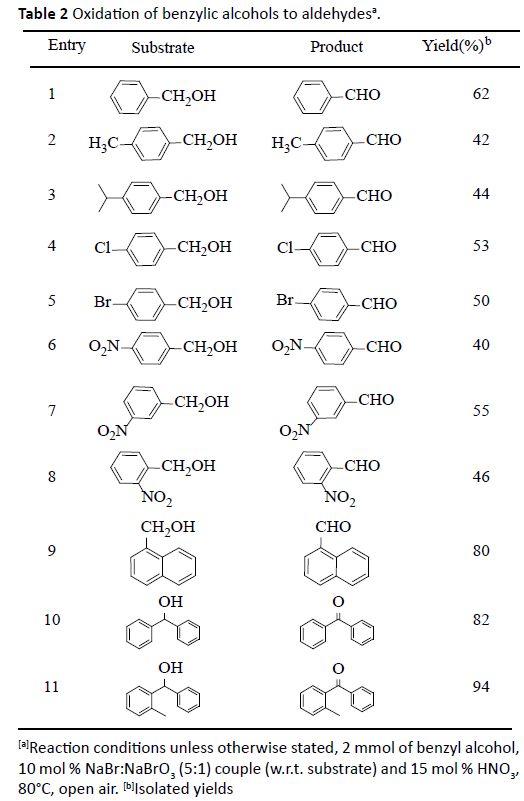
The electronic effect and substitution pattern of substituents on the phenyl ring of benzyl alcohol has profound effect on the reaction rates and product yield (Table 2). In general, electronic releasing alkyl group (entry 2 and 3) as well as mild electronic withdrawing chloro and bromo (entries 4 and 5) derivative at para position gave moderate to good yields of corresponding aldehydes (42- 53%). Highly electron withdrawing nitro groups were react slowly and leads to moderate to good yields (entries 6-8). 1-Napthalene methanol was also converted to 1-naphthaldehyde in 80% yield (entry 9). Noteworthy to mention that, secondary alcohols like diphenylmethonol and phenyl(o-tolyl) methanol are smoothly underwent to oxidation and afforded the corresponding ketones in 82% and 94% yields (entry 10 and 11). This might be due to benzylic effects of two benzene rings to stabilizes a reaction intermediates at transition states.
Mechanism
Based on experimental observations and literature reports [12], we proposed a probable mechanism for the present transformation (Scheme 1). The bromide-bromate and acid (H+) generates bromine in-situ which under aqueous conditions disproportionate to HBr and BrOH. BrOH is revealed to be a mild and selective oxidizing agent for a number of organic substrates [11,12]. BrOH is the primary oxidant and oxidizes alcohol to aldehyde with the elimination of HBr (Scheme 1). The eliminated HBr is oxidized by HNO3 to regenerate BrOH under aqueous conditions as predicted in eqs. 1-7. However, it should be noted that HNO3 and O2 (atmosphere) are necessary as secondary oxidants to complete the catalytic cycle [50-52].
Conclusion
In conclusion, we have demonstrated a simple, economical and sustainable route for selective oxidation of alcohol to corresponding aldehydes and ketones under mild reaction conditions. The yields obtained were moderate to good, compared to known oxidation systems, the present protocol is more sustainable. As both the bromide/bromate couple (“Br”) and HNO3 are inexpensive, easily available even in a undergraduate chemical laboratory. In addition, the use of atmosphere oxygen as co-oxidant makes the entire process attractive from green chemistry point of view.
Acknowledgements
CSIR-CSMCRI Communication No. 145/2016. VP is thankful to UGC, New Delhi, India for his fellowship. We thank CSIR-CSMCRI (OLP-0087 and MLP-0027) for financial support.
References
- Sheldon RA, Kochi JK (1981) Metal catalysed oxidations of organic compounds. Academic Press, New York, USA.
- Okada T, Asawa T, Sugiyama Y, Iwai T, Kirihara M, et al. (2016) Sodium hypochlorite pentahydrate (NaOCl·5H2O) crystals; An effective re-oxidant for TEMPO oxidation. Tetrahedron 72: 2818-2827.
- Verma S, Baig RN, Nadagouda MN, Varma RS (2016) Selective oxidation of alcohols using photoactive VO@ g-C3N4. ACS Sustainable Chemistry & Engineering 4: 1094-1098.
- Yamazaki SI, Yao M, Fujiwara N, Siroma Z, Yasuda K, et al. (2012) Electrocatalytic oxidation of alcohols by a carbon-supported Rh porphyrin. Chemical Communications 48: 4353-4355.
- Kuang Y, Islam NM, Nabae Y, Hayakawa T, Kakimoto MA (2010) Selective aerobic oxidation of benzylic alcohols catalyzed by carbon-based catalysts: a nonmetallic oxidation system. Angewandte Chemie International Edition 49: 436-440.
- Park HJ, Lee JC (2009) Oxidation of benzylic alcohols with urea-hydrogen peroxide and catalytic magnesium bromide. Syn Lett 1: 79-80.
- Hoover JM, Ryland BL, Stahl SS (2013) Copper/TEMPO-catalyzed aerobic alcohol oxidation: mechanistic assessment of different catalyst systems. ACS Catalysis 3: 2599-2605.
- Steves JE, Stahl SS (2015) Stable TEMPO and ABNO Catalyst Solutions for User-Friendly (bpy) Cu/Nitroxyl-Catalyzed Aerobic Alcohol Oxidation. The Journal of Organic Chemistry 80: 11184-11188.
- Xie S, Tsunoyama H, Kurashige W, Negishi Y, Tsukuda T (2012) Enhancement in aerobic alcohol oxidation catalysis of Au25 clusters by single Pd atom doping. ACS Catalysis 2: 1519-1523.
- Costa VV, Estrada M, Demidova Y, Prosvirin I, Kriventsov V (2012) Gold nanoparticles supported on magnesium oxide as catalysts for the aerobic oxidation of alcohols under alkali-free conditions. Journal of Catalysis 292: 148-156.
- Wang L, Li J, Lv Y, Zhao G, Gao S (2012) Selective aerobic oxidation of alcohols catalyzed by iron chloride hexahydrate/TEMPO in the presence of silica gel. Applied Organometallic Chemistry 26: 37-43.
- Hollmann F, Arends IW, Buehler K, Schallmey A, Bühler B (2011) Enzyme-mediated oxidations for the chemist. Green Chemistry 13: 226-265.
- Shaabani A, Laeini MS, Shaabani S, Seyyedhamzeh M (2016) NaBrO3/guanidinium-based sulfonic acid: as a transition metal-and strong inorganic acid-free oxidation system for alcohols and alkyl arenes. New Journal of Chemistry 40: 2079-2082.
- Gazi S, Ananthakrishnan R (2012) Bromodimethylsulfonium bromide as a potential candidate for photocatalytic selective oxidation of benzylic alcohols using oxygen and visible light. RSC Advances 2: 7781-7787.
- Uyanik M, Fukatsu R, Ishihara K (2010) Bromine-Catalyzed Aerobic Oxidation of Alcohols. Chemistry–An Asian Journal 5: 456-460.
- Paakkonen S, Pursiainen J, Lajunen M (2010) Fast oxidation of secondary alcohols by the bromate-bromide system using cyclic microwave heating in acidic water. Tetrahedron Letters 51: 6695-6699.
- Podgorsek A, Stavber S, Zupan M, Iskra J (2007) Bromination of ketones with H2O2–HBr “on water”. Green Chemistry 9: 1212-1218.
- Hirashima SI, Itoh A (2007) Aerobic oxidation of alcohols under visible light irradiation of fluorescent lamp. Green Chemistry 9: 318-320.
- Sugai T, Itoh A (2007) Aerobic oxidation under visible light irradiation of a fluorescent lamp with a combination of carbon tetrabromide and triphenyl phosphine. Tetrahedron Letters 48: 9096-9099.
- Joseph JK, Jain SL, Sain B (2006) Oxidation of Alcohols and vic-Diols with H2O2 by Using Catalytic Amounts of N-Methylpyrrolidin-2-one Hydrotribromide. European Journal of Organic Chemistry 3: 590-594.
- Hirashima SI, Hashimoto S, Masaki Y, Itoh A (2006) Aerobic photo-oxidation of alcohols in the presence of a catalytic inorganic bromo source. Tetrahedron 62: 7887-7891.
- Kuwabara K, Itoh A (2006) Facile aerobic photooxidation of alcohols in the presence of catalytic N-bromosuccinimide. Synthesis 12: 949-1952.
- Ramachandraiah G, Ghosh PK, Adimurthy S, Mehta AS, Jethva AD, et al. (2004) US Patent No. 6,740,253. Dated 25 May 2004.
- Ramachandraiah G, Ghosh PK, Adimurthy S, Bedekar AV, Shukla DB (2008) US Patent 7,459,139. Dated 02 December 2008.
- Adimurthy S, Ramachandraiah G, Bedekar AV, Ghosh S, Ranu BC, et al. (2006) Eco-friendly and versatile brominating reagent prepared from a liquid bromine precursor. Green Chemistry 8: 916-922.
- Adimurthy S, Ghosh S, Patoliya PU, Ramachandraiah G, Agrawal M, et al. (2008) An alternative method for the regio-and stereoselective bromination of alkenes, alkynes, toluene derivatives and ketones using a bromide/bromate couple. Green Chemistry 10: 232-237.
- Dohi T, Fukushima KI, Kamitanaka T, Morimoto K, Takenaga N, et al. (2012) An excellent dual recycling strategy for the hypervalent iodine/nitroxyl radical mediated selective oxidation of alcohols to aldehydes and ketones. Green Chemistry 14: 1493-1501.
- Li XQ, Zhang C (2009) An environmentally benign TEMPO-catalyzed efficient alcohol oxidation system with a recyclable hypervalent iodine (III) reagent and its facile preparation. Synthesis 07: 1163-1169.
- Zhao XF, Zhang C (2007) Iodobenzene dichloride as a stoichiometric oxidant for the conversion of alcohols into carbonyl compounds; Two facile methods for its preparation. Synthesis 04: 551-557.
- Vatèle JM (2006) Yb (OTf) 3-Catalyzed oxidation of alcohols with iodosylbenzene mediated by TEMPO. Syn Lett 13: 2055-2058.
- Mu R, Liu Z, Yang Z, Liu Z, Wu L, et al. (2005) An efficient catalytic aerobic oxidation of alcohols in water using hypervalent iodine (V). Advanced Synthesis & Catalysis 347: 1333-1336.
- Zhang S, Xu L, Trudell ML (2005) Selective oxidation of benzylic alcohols and TBDMS ethers to carbonyl compounds with CrO3-H5IO6. Synthesis 11: 1757-1760.
- Gogoi P, Konwar D (2005) Transition-metal-and organic-solvent-free: a highly efficient anaerobic process for selective oxidation of alcohols to aldehydes and ketones in water. Organic & Biomolecular Chemistry 3: 3473-3475.
- Krishnaveni NS, Surendra K, Rama Rao K (2004) A Simple and Highly Selective Biomimetic Oxidation of Alcohols and Epoxides with N-Bromosuccinimide in the Presence of ß-Cyclodextrin in Water. Advanced Synthesis & Catalysis, pp: 346-350.
- Uyanik M, Akakura M, Ishihara K (2009) 2-Iodoxybenzenesulfonic acid as an extremely active catalyst for the selective oxidation of alcohols to aldehydes, ketones, carboxylic acids, and enones with oxone. J Am Chem Soc 131: 251-262.
- Noyori R (2005) Pursuing practical elegance in chemical synthesis. Chemical Communications 14: 1807-1811.
- Huang Z, Li F, Chen B, Yuan G (2015) Sustainable catalytic oxidation of alcohols over the interface between air and water. Green Chemistry 17: 2325-2329.
- Assady E, Yadollahi B, Riahi Farsani M, Moghadam M (2015) Zinc polyoxometalate on activated carbon: an efficient catalyst for selective oxidation of alcohols with hydrogen peroxide. Applied Organometallic Chemistry 29: 561-565.
- Chen T, Cai C (2015) Selective Oxidation of Benzyl Alcohols to Aldehydes with a Salophen Copper (II) Complex and tert-Butyl Hydroperoxide at Room Temperature. Synthetic Communications 45: 1334-1341.
- Liu C, Fang Z, Yang Z, Li Q, Guo S, et al. (2015) Highly practical sodium (I)/azobenzene catalyst system for aerobic oxidation of benzylic alcohols. RSC Advances 5: 79699-79702.
- Joshi G, Patil RD, Adimurthy S (2012) Green bromine: in situ, generated catalyst for the selective oxidation of alcohols using H2O2 as a benign oxidant. RSC Advances 2: 2235-2239.
- Samanta S, Pappula V, Dinda M, Adimurthy S (2014) Transition metal-free oxidative esterification of benzylic alcohols in aqueous medium. Organic & Biomolecular Chemistry 12: 9453-9456.
- Pappula V, Adimurthy S (2016) Green process development for the preparation of 2, 6-dibromo-4-nitroaniline from 4-nitroaniline using bromide–bromate salts in an aqueous acidic medium. RSC Advances 6: 90184-90187.
- Pappula V, Donthiri RR, Patil RD, Adimurthy A (2013) S Journal of Energy and Chemical Engineering 1: 27-50.
- Patil RD, Adimurthy S (2011) Copper-Catalyzed Aerobic Oxidation of Amines to Imines under Neat Conditions with Low Catalyst Loading. Advanced Synthesis & Catalysis 353: 1695-1700.
- Joshi G, Adimurthy S (2011) Environment-Friendly Bromination of Aromatic Heterocycles Using a Bromide–Bromate Couple in an Aqueous Medium. Industrial & Engineering Chemistry Research 50: 12271-12275.
- Joshi G, Bhadra S, Ghosh S, Agrawal MK, Ganguly B, et al. (2009) Making full use of the oxidizing equivalents in bromate in the selective oxidation of thiols, sulfides, and benzylic/secondary alcohols into disulfides, sulfoxides, and aldehydes/ketones. Industrial & Engineering Chemistry Research 49: 1236-1241.
- Patil RD, Joshi G, Adimurthy S (2010) HBr- H2O2: A Facile Protocol for Regio-selective Synthesis of Bromohydrins and a-Bromoketones and Oxidation of Benzylic/Secondary Alcohols to Carbonyl Compounds under Mild Aqueous Conditions. Industrial & Engineering Chemistry Research 49: 8100-8105.
- Patil RD, Bhadra S, Adimurthy S, Ranu BC (2010) Green Oxidation of Methylarenes to Benzoic Acids with Bromide/Bromate in Water. Synthetic Communications 40: 2922-2929.
- Ross DS, Gu CL, Hum GP, Malhotra R (1986) Oxidation of benzyl alcohols by nitrous and nitric acids in strong sulfuric acid media. International Journal of Chemical Kinetics 18: 1277-1288.
- Strazzolini P, Runcio AE (1990) J Org Chem 203: 523-526.
- Joshi SR, Kataria KL, Sawant SB, Joshi JB (2005) Kinetics of oxidation of benzyl alcohol with dilute nitric acid. Industrial & Engineering Chemistry Research 44: 325-333.
Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences