Fungus and their Anti-Cancerous Properties
Sujoy Pal1*, Nobendu Mukerjee1, Asmita Ghosh2, Nwiboko Michael Chukwuemeka3
Sujoy Pal1*, Nobendu Mukerjee1, Asmita Ghosh2, Nwiboko Michael Chukwuemeka3
1Department of Microbiology, Ramakrishna Mission Vivekananda Centenary College, Kolkata, India
2Department of Biotechnology, Heritage Institute of Technology, Kolkata, India
3Department of Applied Microbiology, Ebonyi State University, Kolkata, India
- *Corresponding Author:
- Sujoy Pal
Department of Microbiology
Ramakrishna Mission Vivekananda Centenary College
Kolkata, India
Tel: 7044326745
E-mail: sujoypal2005@yahoo.co.in
Received Date: May 16, 2021; Accepted Date: September 02, 2021; Published Date: September 13, 2021
Citation: Sujoy P (2021) Fungus and their Anti-Cancerous Properties. J Org Inorg Chem Vol.07 No.05.
Abstract
Fungus are a valuable source of compounds of therapeutic relevance, and it has many cytotoxic effects and to be a bit precise of anticancer compounds [1]. Furthermore, herbal product’s overall synthesis is a treasured device most effective for the development of original complex biologically active compounds as well as for the improvement of innovative methodologies in enantio- selective natural synthesis [2]. Natural products play a vital role in the development of anticancer drugs. Till date, predominantly metabolites from fungi served as lead structures for anticancer agents. Fungal metabolites and their derivatives are much less investigated for their potential in cancer therapy. This review gives a detailed overview on the role of fungal derivatives in cancer therapy and summarizes some of the latest results of our group in this area.
Keywords
Fungal anticancer metabolites; biological activity; drug discovery; mechanism of action
Introduction
Various fungal metabolites have been proven for anticancerous properties both in vitro and in animal models and some synthetic analogs of those metabolites have progressed into clinical trials [2]. Currently, there are very few fungal agents that have been clinically proven as potential anticancer drugs. Two probable reasons could be envisioned:
• Lack of clear understanding of anticancer mechanism of action in fungi
• Unable to supply enough materials to support the preclinical and clinic developments.
In this review, we will summarize the sources and mechanisms of action of some of these fungal metabolites that have numerous promising anticancer effects. We aim to evaluate the anti-cancerous properties of fungal metabolites, with importance on their mode of action, thus increasing the possibility of pioneering artificial procedures that assist the progress of fungal metabolites into drugs that might have future implications.
Materials and Methods
Detailed analysis with respect to each fungus and their characteristics has been used as a baseline tool to guide our review study. A proper knowledge regarding the mechanism of action related to each fungus’s anti-cancerous properties were considered before writing the review paper. Their etiology and molecular and genetic properties were analyzed also.
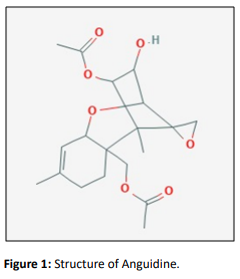
Anguidine
Anguidine is also known as 4, 15-diacetoxyscirpenol, is a known mycotoxin belonging to the group of trichothecenes produced by several Fusarium species, including F. roseum, F. sambucinum, F. tricinctum, F. equiseti, F. l ateritium, F. graminarium, F. semitectum, F. sulfureum, F. diversis porum, F. scirpi, and Giberellaintrigans [4].
Mechanism of action
Anguidine inhibits synthesis of protein by arresting the protein chain initiation through an irreversible process of degradation of polyribosomes [5]. Trichothecenes are toxic metabolites responsible for a cellular division like intestinal crypt epithelium and hematopoietic cells. Cytotoxicity has been coupled with protein impairment or cellular membrane dysfunction. The inhibition of protein synthesis has been associated with the insertion of labile and some regulatory proteins like IL-2 in immunocytes. Small molecule transportation is hindered within the cell membranes due to extremely low concentrations of trichothecenes. Early preclinical studies at the National Cancer Institute (NCI) revealed >100% life-span extension of P388 leukemia-bearing mice with chronic i.e. administration of anguidine. No significant activity was established in six tumor categories according to the Phase II study conducted at M. D. Anderson Hospital and Tumor Institute and the University of Kansas (the Southeastern Cancer Study Group) and thus, it was concluded that anguidine did not warrant additional evaluation for the treatment of solid tumors [4, 5].
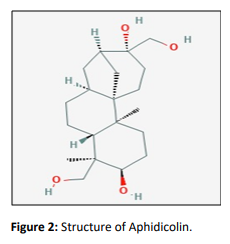
Aphidicolin
Aphidicolinis a diterpenoid metabolite produced by different fungi including Cephalosporium aphidicola, Nigrospora sphaerica, and Harziella entomophilla. Its structure was determined using chemical and spectroscopic data and later confirmed by its stereocontrolled synthesis [6]. More recently, aphidicolin was also isolated from Phoma sp. 7210 associated with Aizooncanariense. Prominent researches show that Aphidicolin kills neuroblastoma cells selectively in vitro by binding to alpha DNA polymerase and inhibiting cell growth.It is important because of its anti-mitotic and anti-mitotic activity that acts as a potent inhibitor of DNA replication in various organisms [7].
Mechanism of action
Aphidicolin inhibits theα and δ chain of DNA polymerases. DNA polymerases α and δ inhibition by aphidicolin is reversible. Thus, it has been widely used as a synchronizing agent in various experimental setups. Its water-soluble analogue aphidicolin glycinate underwent Phase I clinical trials, and established a dosage after24-hour continuous infusion as the recommended schedule for clinical evaluations of aphidicolin glycinate. It established itself as a synchronizing agent in combination with cisplatin [8].The inhibitor blocks the attachment of dCTP by docking at the Pol α active site by revolving the guanine model. The structure provides conceivable machinery for the selectivity of aphidicolin incorporation across template guanine. This helps in understanding the previous modifications of aphidicolin and its reason for failure in the improvement of its affinity towards Pol α [7].
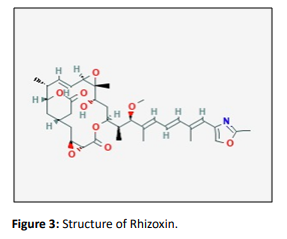
Rhizoxin
Rhizoxin is a phytotoxic 16-membered macrolide, initially isolated from the cultures of the fungus Rhizopuschinensis, the causal agent of rice seedling blight [9].The spectroscopic and several biochemical assays helped in Structural determination. Successive studies proved this potent antimycotic polyketide not to be produced by the fungus itself, but rather by endosymbiotic bacteria belonging to the Burkholderia genus, among which the best producer appeared to be Burkholderia rhizoxina. Rhizoxin binds to tubulin situated at the vinca site and hinders the microtubule assembly [6].More recently, a combination of genetic and chemical studies allowed ascertaining that the macrolide is first epoxidized by the bacteria cytochrome P450 monooxygenase RhiHand that the 2, 3-oxirane rings are then introduced by the fungal host to specifically tailor the rhizoxin scaffold. The additional epoxide moiety significantly increases phytotoxic effectiveness. The large-scale fermentation of Rhizoin led to the production and identification of a hefty number of Rhizoxin natural analogues [10, 11].
Mechanism of action
Rhizoxin binds to tubulin at the Vinca site, thereby inhibiting microtubule assembly. It attaches to the beta- tubulin in eukaryotic cells which disrupts microtubule formation [11]. Hence, it prevents formation of the mitotic spindle inhibiting cell division. Additionally, rhizoxin can depolymerize assembled microtubules. The purpose of Rhizoxin is much alike to some alkaloids like Vinca which was also found to show some cytotoxic effects against some variety of human tumor cells and some xenograft models [12]. On completion of phase I, some clinical trials provided some maximum dosage of the compounds; low leveled toxicity. At phase II plethora of clinical trials were done along with them EROTC early clinical trials group focused on treatments of cancer. These too include melanoma, thoracic cancer, and NSCLC [13, 14]. It was later concluded at melanomas trial that these patients became tolerant to scheduled dosage, hence no severe change and sign of improvement at health of patient was seen. In the NSCLC trial, better results were observed, thereby supporting Rhizoxin as a promising anticancer therapeutic tool. This consisted of 4 partial responses and almost 50% of the patients treated showed stabilization of the disease condition. This led to the conclusion that rhizoxin showed activity as a single agent, and its application to NSCLC treatment paved a way for further research and evaluation [15,16, 17, 18].
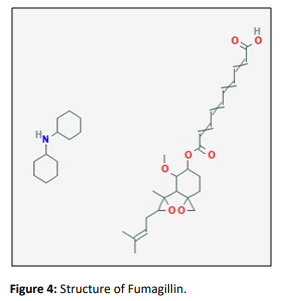
Fumagillin
Fumagillin is a complex metabolite first isolated from the liquid culture of Aspergillus fumigatusstrain H-3. Its structure was assigned based on chemical studies which included the preparation of a number of key derivatives of interest. Biosynthetic studies showed that fumagillin arises by a terpene route and acetate pathway in parts.Fumagillin was also produced by other species of Aspergillus, including A. flavus and A. parasiticus. Recently, a chlorinated derivative of fumagillin (ligerine) was isolated from a marine-derived Penicillium strain, showing strong inhibitory activity against an osteosarcoma cell line.
Mechanism of action
Synthetically derived fumagillin analogues, TNP-470, PPI-2458 and CKD-732, have been evaluated in human cancer clinical trials. Together with fumagillin, these synthetic analogues disrupt tumor vasculature by targeting the enzyme methionine aminopeptidase type 2, which cleaves the N-terminal methioninol residue of newly synthesized proteins. TNP470 is a fumagillin analogue that was isolated from an infected culture of capillary endothelial cells originally. Purified fumagillin exhibits inhibition of endothelial cell proliferation in in vitro conditions and tumor induced angiogenesis in vivo conditions. Fumagillin aids in blood vessel blockage by binding to the enzyme methionine aminopeptidase 2. For this reason, this metabolite, in conjugation with semisynthetic derivatives, are studied as an angiogenesis inhibitor in cancer therapeutics. Although promising results for TNP-410 were reported in patients with prostate cancer, NSCLC and melanoma, neurotoxicity was observed to be an important side-effect, seemingly limiting its clinical usefulness. Mechanism to neurotoxicity and growing strategies to irradiate the dose limiting and some side effects that would be required ahead this agent could be widely used at clinics. Newly PPI2456 & CKD732 appeared to take the edge of neurotoxical issues coupled with TNP470.Its therapeutic effects remain to be demonstrated in the clinic. [19]
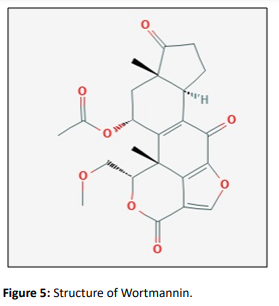
Wortmannin
Wortmannin is a furan steroid metabolite isolated from Penicillium wortmannin. Wortmannin was also isolated from Fusarium torulosum and Trichoderma sp. It has a promising use in anti-cancer therapy as an apoptosis-based therapeutic agent in the treatment of cancers. (W.Kohn, 2002). Treatment with wortmannin blocks the DNA-damage-induced p53 stabilization and results in radioresistant DNA synthesis. This knowledge helps in the efficient management and treatment of cancers based on specific alterations that are used in a tumor [20]
Mechanism of action
This fungal metabolite is inhibitor DNA dependent kinases like phosphatidylinositol 3′ kinases (PI3Ks) and phosphatidylinositol 3′ kinase-related kinases (PIKKs).These inhibitors target the kinase domain of the PI3Ks and which hinder their kinase activity [21]. Wortmann forms covalent interactions with a critical lysine residue in the ATP-binding site of p110 kinase domains. This prevents ATP binding and transfer of the gamma phosphate group from ATP to PIP2 when generating PIP3. PX-866 is the most successful modification of Wortmannin, with enhanced in vivo stability and favorable efficacy in both cells and mouse models of cancer [22]. PI3ks inhibition process the opportunity to resist a vast cancer cell line to survive its signaling pathway and to overcome the activity of expunged Tumor suppressor which provides an anti tumouric property with increased sensitivity to tumor to a spacious diversity of drugs. Preclinical studies exposed potential radio sensitizing activity connected with Wortmannin. However, side effects like high toxicity, poor stability, and low water solubility hindered its clinical translations. A large number of analogues have been developed to overcome wortmannin's poor pharmacokinetic properties till now. PX-866 is currently under clinical evaluation. [23]
Meleagrin
Meleagrin is an indole alkaloid compound derived from endop hytic fungus Penicillium chrysogenum indigenously present in the leaf of the olive tree Olea Europa in Egypt. This fungal metabolite shows promising inhibitory activity against the proliferation of human breast cancer cell lines likeMDA-MB-231, MDA-468, BT-474, SK BR-3, MCF7 and MCF7dox. Melagrin B contains a rare diterpene moiety that exhi bited potent Cytotoxicity against diverse cell lines like A-549, HL-60, BEL-7402, and MOLT-4[24].
Mechanism of action
Z-lyte assay of Meleagrin showed significant wild and mutant c-Met inhibitory activity [25]. Meleagrin dosage showed a significant decline of the activated (phosphorylated) c-Met level with no effects on the total Met level as observed in western blot analysis of MDA-MB-23 cells [26]. Meleagrin also displayed anti-proliferative activity in breast cancer cell lines. ERα (Estrogen receptor α) is expressed in MCF7, MCF7-dox and BT-474 cells, whereas MDA-MB-231 and MDA-MB-468 cells lack the expression of ERα [25]. Meleagrin treatment showed positive inhibitory regulation of the invasive triple negative breast tumor cell growth in an orthotopic athymic nude mice model. The indole alkaloid meleagrin is a novel lead c-Met inhibitory entity that controls c-Met-dependent metastatic and invasive breast malignancies. These results also proved that inhibitory effects on the c-Met signaling are linked with breast cancer proliferation through the effects of Meleagrin. One of the mechanisms by which c-Met signaling dysfunction leads to cancer proliferation is through the gain of function mutations in the c-Met proto oncogenes which result in the alter of amino acid substitutions in the tyrosine kinase and in the juxtamembrane domains of the c-Met [26].
Conclusion
The advent of high-throughput screens involving molecular targets created a demand for the generation of large combinatorial libraries of compounds, the shift away from purely synthetic to more natural product-like compounds, either derived from or inspired by natural products, has continued to work its way back to drug discovery programs. It has been now generally recognized that the hit rates obtained using the natural product-derived collections of compounds are significantly higher than those resulting from combinatorial libraries [27, 28]. Furthermore, the new understanding of Lipinski’s rules emphasizes that they have to be applied with caution to natural products, which are recognized by active transport systems [29]. Thus, natural products which have already proved their worth in many areas of human medicine, and particularly in cancer drug discovery, are expected to draw an increasing amount of attention yet again. Although a fungal metabolite is yet to lead to an approved anticancer drug, the examples in the current review demonstrate that the tremendous structural diversity and promising anticancer potential of many of these natural products will eventually lead to this desired breakthrough.
References
- Hanahan D, Weinberg RA (2011) Hallmarks of cancer: the next generation. Cell 144: 646–674.
- Barbero M, Artuso E, Prandi C (2018). Fungal Anticancer Metabolites: Synthesis Towards Drug Discovery. Current Medicinal Chemistry 25(2).
- Kanoh K, Kohno S, Asari T (1997) (−)-Phenylahistin: a new mammalian cell cycle inhibitor produced by Aspergillusustus. Bioorg Med Chem Lett 7: 2847–2852.
- Kornienko A, Evidente A, Vurro M, Mathieu V, Cimmino A, Evidente M, Kiss R (2015) Toward a Cancer Drug of Fungal Origin. Medicinal Research Reviews 35(5): 937–967.
- Yamori T (1997) A human cell line panel for screening anti-cancer drugs. Gan To Kagaku Ryoho 24: 129–135.
- Kornienko A, Evidente A, Vurro M, Mathieu V, Cimmino A, Evidente M, Kiss R (2015) Toward a Cancer Drug of Fungal Origin. Medicinal Research Reviews 35(5): 937–967.
- Baranovskiy AG, Babayeva ND, Suwa Y, Gu J, Pavlov YI, Tahirov TH (2014) Structural basis for inhibition of DNA replication by aphidicolin. Nucleic Acids Research 42(22): 14013–14021.
- Eliza Starczewska, Maxime Beyaert, Lucienne Michaux, Marie-Christiane Vekemans, Pascale Saussoy, Vanesa Bol, Ainhoa Arana Echarri, Caroline Smal, Eric Van Den Neste, Françoise Bontemps (2016) Targeting DNA repair with aphidicolin sensitizes primary chronic lymphocytic leukemia cells to purine analogs; Oncotarget 7(25): 38367–38379.
- Rath CM, Scaglione JB, Kittendorf JD, Sherman DH (2010) NRPS/PKS Hybrid Enzymes and Their Natural Products. Comprehensive Natural Products II 453–492.
- Hearn BR, Shaw SJ, Myles DC (2007) Microtubule Targeting Agents. Comprehensive Medicinal Chemistry II 81–110.
- Takahashi M, Iwasaki S, Kobayashi H (1987) Studies on macrocyclic lactone antibiotics. XI. Anti-mitotic and anti-tubulin activity of new antitumor antibiotics, rhizoxin and its homologues. J Antibiot (Tokyo) 40: 66–72.
- Takahashi M, Iwasaki S, Kobayashi H et al (1987) Rhizoxin binding to tubulin at the maytansine-binding site. BiochimBiophysActa 926: 215–223.
- Tsuruo T, Oh-hara T, Iida H (1986) Rhizoxin, a macrocyclic lactone antibiotic, as a new antitumor agent against human and murine tumor cells and their vincristine-resistant sublines. Cancer Res 46: 381–385.
- Roberge M, Cinel B, Anderson HJ (2000) Cell-based screen for antimitotic agents and identification of analogues of rhizoxin, eleutherobin, and paclitaxel in natural extracts. Cancer Res 60: 5052–5058.
- Hendriks HR, Plowman J, Berger DP (1992) Preclinical antitumour activity and animal toxicology studies of rhizoxin, a novel tubulin-interacting agent. Ann Oncol 3: 755–763.
- Arioka H, Nishio K, Heike Y (1997) In vitro and in vivo modulation by rhizoxin of non-P-glycoprotein-mediated vindesine resistance. J Cancer Res Clin Oncol 123: 195–200.
- Kurihara A, Shibayama Y, Mizota A (1996) Enhanced tumor delivery and antitumor activity of palmitoyl rhizoxin using stable lipid emulsions in mice. Pharm Res 13: 305–310.
- Tolcher AW, Aylesworth C, Rizzo J (2000) A phase I study of rhizoxin (NSC 332598) by 72-hour continuous intravenous infusion in patients with advanced solid tumors. Ann Oncol 11: 333–338.
- Zhang Y, Yeh JR, Mara A, Ju R, Hines JF, Cirone P, Crews CM (2006) A Chemical and Genetic Approach to the Mode of Action of Fumagillin. Chemistry & Biology 13(9): 1001–1009.
- Wymann MP, Bulgarelli-Leva G, Zvelebil MJ, Pirola L, Vanhaesebroeck B, Waterfield MD, Panayotou, G (1996) Wortmannin inactivates phosphoinositide 3-kinase by covalent modification of Lys-802, a residue involved in the phosphate transfer reaction. Molecular and Cellular Biology 16(4): 1722–1733.
- Sheridan C, Downward J (2013) Inhibiting the RAS–PI3K Pathway in Cancer Therapy. The Enzymes 107–136.
- Fujita K, Ishikawa F, Kakeya H (2014) Biosynthetic origins of the epoxyquinone skeleton in epoxyquinols A and B. J Nat Prod 77: 2707–2710.
- Zhu T, Gu J, Yu K, Lucas J, Cai P, Tsao R, Zask A (2006) Pegylated Wortmannin and 17-Hydroxywortmannin Conjugates as Phosphoinositide 3-Kinase Inhibitors Active in Human Tumor Xenograft Models. Journal of Medicinal Chemistry 49(4): 1373–1378.
- Elnagar A, Sylvester P, El Sayed K (2011) (−)-Oleocanthal as a c-Met Inhibitor for the Control of Metastatic Breast and Prostate Cancers. Planta Medica 77(10): 1013–1019.
- Wang W, Marimuthu A, Tsai J, Kumar A, Krupka HI, Zhang C, West BL (2006) Structural characterization of autoinhibited c-Met kinase produced by coexpression in bacteria with phosphatase. Proceedings of the National Academy of Sciences 103(10): 3563–3568.
Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences